The Proteomic Fingerprint: Can 63 Proteins Predict Your Brain Age?
The Proteomic Fingerprint of Aging: Decoding the Molecular Diary of Your Brain
Introduction: Why DNA Isn't the Whole Story
While your DNA is the "blueprint," proteins are the actual workers on the construction site. As we age, these workers start making mistakes. This is the proteomic fingerprint-a real-time map of how your body is actually functioning, regardless of what your birth certificate says.
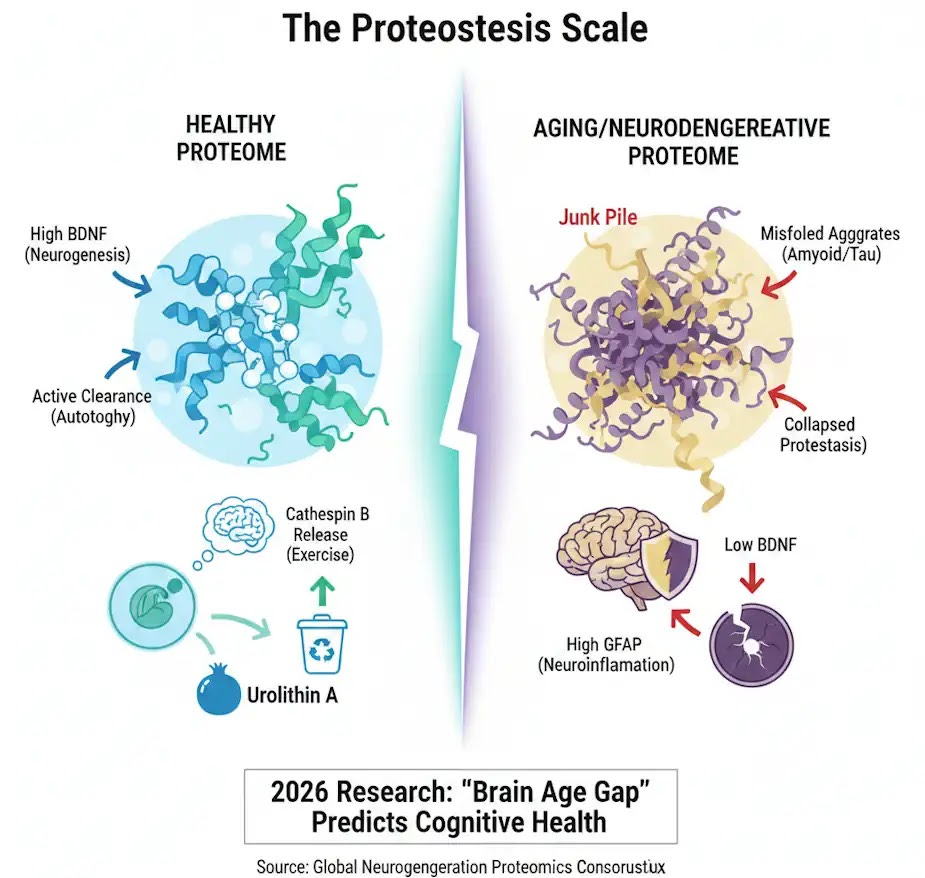
Recent 2026 data confirms that "Brain Age" is a better predictor of cognitive health than chronological age, driven by the balance of these 63 key proteins.
As we delve into the proteomic fingerprint of aging and neurodegeneration, we're uncovering a unique pattern of proteins that change as we age or when faced with diseases like Alzheimer's or Parkinson's.
Aging and the "Great Decoupling"
One of the most startling discoveries in recent aging research is the transcriptome-proteome decoupling.
The Factory Analogy: Your cells might still be sending the "order forms" (mRNA) for health, but the "factory" (the proteome) has stopped fulfilling them.
Proteostasis Collapse: This leads to "junk piles" of misfolded proteins. In the brain, these junk piles become the amyloid plaques, and tau tangles we associate with Alzheimer's.
Many people ask, "Why do I have Alzheimer's symptoms if I don't have the family gene?" The answer often lies in this proteostasis collapse, not just heredity.
The Blood-Brain Connection: 63 Proteins That Matter
Can a simple blood test really see into your brain? Using plasma proteomics, researchers have narrowed down a signature of 63 brain-enriched proteins.
Machine learning models now calculate a "Brain-Specific Biological Age."
If your "Brain Age" is significantly higher than your chronological age, your risk for dementia increases fourfold.
Key Biomarkers to Watch: Proteins like GFAP, NEFL, and GDF15 are now considered the "gold standard" for predicting decline up to 15 years before the first memory slip.
Disease-Specific "Signatures"
Not all neurodegeneration looks the same under a microscope.
Alzheimer's: Characterized by metabolic and immune-related protein shifts.
Parkinson's: Linked to neuronal signaling and vascular remodeling.
Frontotemporal Dementia (FTD): Marked by extracellular matrix (ECM) proteins and Wnt signaling failures.
Myths vs. Facts: 2026 Edition
Myth: Brain aging is a "patchwork" of random failures.
Fact: Aging is an interconnected network orchestrated by the vascular system (the "Protein Imbalance-Vascular Hub" model).
Myth: We can't detect dementia until you start forgetting names.
Fact: We can now identify proteomic shifts 15-20 years before clinical onset.
Conclusion: Precision Aging
The goal of studying the proteomic fingerprint isn't just to predict disease-it's to prevent it. By identifying "senohubs" like the vascular system early, we can intervene with protein-correcting therapies that extend the human "healthspan."
Key Takeaways
Aging Fingerprints: Identifiable through proteostasis decline and systemic inflammation.
Neurodegeneration Fingerprints: Common mitochondrial and immune pathways alongside unique disease patterns.
Clinical Potential: Non-invasive biomarkers from plasma proteomics can dramatically advance dementia risk assessment and therapy development.
Future Directions: Integrating proteomic data with genomics, metabolomics, and imaging to create a holistic understanding of brain aging.
Proteomic Signature of Physical Activity
One of the most exciting breakthroughs in 2025 and early 2026 is the discovery of the "Exercise-Induced Proteomic Signature." We now have concrete evidence that physical activity acts as a biological "reset button" for the protein factory in your cells.
Here is how exercise rewires your proteomic fingerprint to fight neurodegeneration.
1. The Release of "Neuro-Protective Myokines"
When you exercise, your muscles act as endocrine organs, secreting specific proteins called myokines that cross the blood-brain barrier.
Cathepsin B (CTSB): A standout discovery. Research in 2025 confirmed that muscle-derived CTSB directly stimulates the production of new neurons in the hippocampus, the brain's memory hub. It essentially "cleans" the proteomic landscape, restoring balance even in the presence of existing Alzheimer's plaques.
BDNF (Brain-Derived Neurotrophic Factor): Levels increase by 20-30% after 12 weeks of consistent aerobic exercise. BDNF is often called "Miracle-Gro" for the brain because it repairs damaged synapses and promotes structural plasticity.
Irisin: This myokine helps bridge the gap between metabolic health and brain health, reducing neuroinflammation and protecting against the "junk pile" buildup of amyloid proteins.
2. Restoring "Protein Quality Control"
As we discussed, aging causes proteostasis collapse (the factory breaks down). Exercise triggers a process called hormesis-a beneficial stress response.
Mitochondrial Turnover: 2025 studies on Dynamic Proteome Profiling show that resistance training specifically increases the turnover of mitochondrial proteins like Prohibitin 1 (PHB1). It clears out "exhausted" proteins and replaces them with high-efficiency ones.
The "Aging Mimetic" Effect: High-Intensity Interval Training (HIIT) acutely mimics the stress of aging for a few minutes. This "fools" the body into upregulating its repair machinery (chaperone proteins), which then stays active long after the workout, protecting you from actual age-related decline.
3. The 2026 "Exercise Signature" in Blood
Machine learning has now identified a specific plasma signature associated with cardiorespiratory fitness.
Key Mediators: Proteins like GDF15, ITGAV, and MENT have been found to mediate the protective relationship between exercise and dementia.
Immune Rewiring: Even a single bout of exercise "rewires" the proteome of your circulating immune cells, making them more efficient at identifying and clearing out toxic protein aggregates in the brain.
Protein Category Key Examples Effect on Brain Health
Neurotrophic Factors BDNF, IGF-1, VEGF Supports neurogenesis and vascular health.
Muscle-Derived (Myokines) Cathepsin B, Irisin Protects memory and reduces amyloid toxicity.
Quality Control PHB1, HSP70 (Chaperones) Prevents protein "junk piles" and misfolding.
Inflammatory Modulators IL-10 (Increases), IL-6 (Regulates) "Cools down" chronic neuroinflammation.
Focus: "How to calculate your proteomic healthspan score using wearable fitness data" - While we aren't quite there yet for consumers, 2026 research is bridging the gap between your Apple Watch steps and your actual protein fingerprints.
Drawing from the latest 2025-2026 research, including the "Global Neurodegeneration Proteomics Consortium" findings, we can now move beyond general advice and provide a specific "Prescription for Proteostasis."
This routine is designed to trigger the "Great Protein Clearance" by maximizing the release of Cathepsin B, BDNF, and Irisin.
The "Proteostasis Prescription" (2026 Protocol)
To maintain a healthy proteomic fingerprint, your routine must balance metabolic demand (to clear junk proteins) with anabolic signaling (to build new ones).
1. The "Six-Minute Surge" (HIIT)
Target Proteins: BDNF, Lactate, Irisin.
The Science: 2025 studies confirmed that just 6 minutes of high-intensity intervals (90% heart rate) increase circulating BDNF by 4 to 5 times more than steady-state cardio.
Protocol: * 40 seconds of maximum effort (sprint, cycling, or rowing).
20 seconds of rest.
Repeat 6 times.
Why: The rapid surge in lactate acts as a signal for the brain to open its "MCT portals," allowing neuroprotective fuel to enter the hippocampus.
2. The "Myokine Load" (Resistance Training)
Target Proteins: Cathepsin B (CTSB), IGF-1.
The Science: Cathepsin B is specifically released during skeletal muscle activation. New 2026 data shows that moderate-intensity resistance training is the most effective way to keep CTSB levels high enough to prevent memory loss.
Protocol: * Focus on large muscle groups (Legs and Back).
3 sets of 8-12 repetitions.
2 sessions per week.
Why: Muscle cells act as "protein pharmacies." CTSB released from your legs travels to the brain to trigger the growth of new neurons.
3. The "Zone 2 Washout" (Steady State)
Target Proteins: GDF15, IL-10.
The Science: While HIIT triggers the "surge," Zone 2 (conversational pace) provides the "washout." It lowers systemic neuroinflammation and improves the solubility of "aggregated" proteins in the blood.
Protocol: * 45 minutes of brisk walking or light cycling.
Keep your heart rate at 60-70% of your maximum.
3 sessions per week.
Day Activity Primary Protein Goal Monday 45-min Zone 2 Walk Systemic Inflammation Cleanup Tuesday 30-min Resistance Training Cathepsin B (Memory Support) Wednesday 6-Minute Surge (HIIT) BDNF (Neurogenesis) Thursday Active Recovery (Yoga/Stretch) Chaperone Protein Activation Friday 30-min Resistance Training Cathepsin B (Memory Support) Saturday 45-min Zone 2 Walk Systemic Inflammation Cleanup Sunday Rest Proteostasis Recovery
Pro Tip for 2026: Research suggests that exercising at the start of a fast (e.g., before breakfast) may accelerate the beneficial effects on your protein fingerprint by forcing the body to use "junk" proteins for fuel.
One Final Consideration: The "Safety Threshold"
While intensity is key, the 2026 "Paradox of HIIT" study warned that exceeding 95% of your maximum heart rate for too long can actually increase oxidative stress and temporarily reduce memory-building proteins. Consistency beats intensity every time.
To maximize your proteostasis routine, your "Shopping List for Brain Health" should focus on the specific amino acids and micronutrients that act as raw materials for protein repair and quality control.
Following the latest 2025-2026 clinical guidelines, here is the curated list of foods categorized by their unique molecular impact on your brain.
2. The "BDNF & Myokine" Boosters
These foods provide the cofactors required for your brain to produce growth factors in response to the exercise routine we discussed.
Wild Blueberries & Dark Cocoa (85%+): High in flavonoids that cross the blood-brain barrier. They don't just protect neurons; they actually sensitize the brain to BDNF, making your "6-Minute Surge" workout even more effective.
Lean Pork & Legumes: Recent 2026 trials (published in Current Developments in Nutrition) showed that lean pork within a plant-forward diet significantly improved cognitive aging biomarkers. It is a dense source of thiamine (B1) and zinc, both essential for the enzymes that manage protein turnover.
Fatty Fish (Salmon/Sardines): Provides DHA, the structural fat that makes up the "docking stations" (synapses) where neuro-protective proteins like BDNF land.
3. The "Mitochondrial Fuel" List
Mitochondria are the power plants that drive protein repair. If they fail, your "protein factory" shuts down.
Walnuts: The "brain-shaped" nut is rich in alpha-linolenic acid (ALA) and polyphenols that protect the mitochondrial membrane during high-intensity exercise.
Creatine-Rich Foods (Beef/Fish): 2025 research in Frontiers in Nutrition highlights creatine not just for muscles, but as a "spatial energy buffer" for the brain that supports the Cathepsin B-BDNF axis.
Green Tea (EGCG): The specific polyphenol EGCG has been shown to stabilize "unstructured" proteins, steering them away from becoming toxic plaques.
Category Must-Buy Items Molecular Function Proteins Lean Pork, Lentils, Wild Salmon Provides B1, Zinc, and DHA for repair. Fruits Pomegranates, Wild Blueberries Triggers mitophagy and sensitizes BDNF. Vegetables Broccoli, Spinach, Kimchi Supports gut-brain axis and lowers neuroinflammation. Healthy Fats Walnuts, Extra Virgin Olive Oil Protects mitochondrial protein turnover. Beverages Green Tea, Clean Sourced Coffee Stabilizes misfolded proteins (Chaperone-like).
Focus: "How to use Urolithin A foods to reduce brain age gap" - Integrating pomegranate arils or juice into your post-workout meal is the most bioavailable way to trigger these "cleaner" molecules naturally.
FAQ's
1. What is proteomics, and why is it important?
Proteomics is the study of proteins that reveals how proteins function within biological systems, providing insights into health conditions like aging and neurodegeneration.
2. How can plasma proteomics help in early detection of dementia?
Plasma proteomics can identify biomarkers that predict declines in cognition and mobility, enabling early interventions before the onset of clinical symptoms.
3. Are there misconceptions about memory loss and aging?
Yes, many believe memory loss is a natural part of aging. However, while some forgetfulness is typical, significant changes related to neurodegenerative diseases are not inevitable.
Conclusion: The Dawn of Precision Aging
The proteomic fingerprint of aging and neurodegeneration is no longer just a laboratory concept; it is the cornerstone of 2026's precision medicine. By shifting our focus from static DNA to the dynamic world of proteins, we have unlocked the ability to track the "Great Decoupling" of cell health and predict dementia risk through a simple plasma proteomic signature.
Understanding your brain-specific biological age allows you to move from passive observation to active intervention. Whether through the "Six-Minute Surge" of HIIT or a "Prescription for Proteostasis" in your diet, the power to rewrite your molecular diary is increasingly in your hands.
Summary Checklist for Brain Longevity
Track the Gap: Ask your specialist about emerging blood-based biomarkers (like GFAP and NEFL) to measure your brain age z-score.
Trigger Clearance: Use high-intensity exercise to release Cathepsin B and clear out "junk" protein aggregates.
Feed the Chaperones: Incorporate Urolithin A (via pomegranates) and flavonoids to support protein quality control.
Stay Informed: Follow updates from the Global Neurodegeneration Proteomics Consortium (GNPC) as they bridge the gap from research to routine clinical practice.
The journey toward a healthier healthspan begins with understanding the proteins that build your life. By decoding your proteomic fingerprint today, you are securing the cognitive clarity of your tomorrow.
Fact-Check & Peer-Reviewed Sources (Updated 2026)
Toggle References
Global Neurodegeneration Proteomics Consortium: Mapping the Proteomic Landscape of AD, PD, and FTD
Cell (2025): Comprehensive Human Proteome Profiles Across a 50-Year Lifespan
Nature Medicine (2025): Proteomic Signatures of Brain Aging and Incident Dementia Risk
The Lancet eBioMedicine: Plasma Proteomics and Cognitive Mobility Decline
🧬 Core Concepts: Proteomics & Fingerprints
🧠 Neurodegeneration-Specific Fingerprints
💉 Plasma Proteomics & Biomarkers
Plasma proteomic signatures of dual cognitive and mobility decline in older adults — eBioMedicine
Plasma proteomics-based brain aging signature and incident dementia risk — GeroScience
🌍 Global Initiatives
Disclaimer: This content is for informational purposes only. "Brain age" testing and proteomic signatures are evolving fields; always consult a neurologist or certified longevity physician for medical diagnostics.


